Sintering refers to the transformation of powdery materials into dense bodies, which is a traditional process.
What is Sintering Technology?
Sintering is the process of pressing and forming solid materials, where heat and pressure are applied to the material, but at a temperature below the melting point of the material. The production of pottery will go through the sintering process, and the process of sintering metal powder into products is called powder metallurgy. After the powder is formed, the dense body obtained by sintering is a polycrystalline material whose microstructure consists of crystals, glass bodies, and pores. The sintering process directly affects the grain size, pore size, and grain boundary shape and distribution in the microstructure, which in turn affects the properties of the material.
The sintering process can occur in natural environments such as the formation of mineral deposits and can be used to manufacture various materials such as metals, plastics, ceramics, etc. The atoms in the material diffuse across the boundaries of the particles, and the particles fuse to form a monolithic solid. Since the temperature of sintering does not reach the melting point of the material, sintering is usually selected as the forming method for materials with extremely high melting points.
Due to the addition of a procedure for making metal powder, the cost of the powder metallurgy process is high, but for alloys such as high melting point, the large difference in melting point of constituent elements, and poor castability, or the melting and casting process cannot be used, but there are high value-added materials. The use of powder metallurgy sintering has the advantages of low process temperature, high sintering compactness, and short sintering time, so it still has quite high competitiveness in addition to the price factor. Using powder sintering technology to develop advanced materials, Japan and European and American countries have actively invested in forward-looking process technology and machine equipment. To enhance the sintering result, an electric current is added to assist in the traditional sintering process.
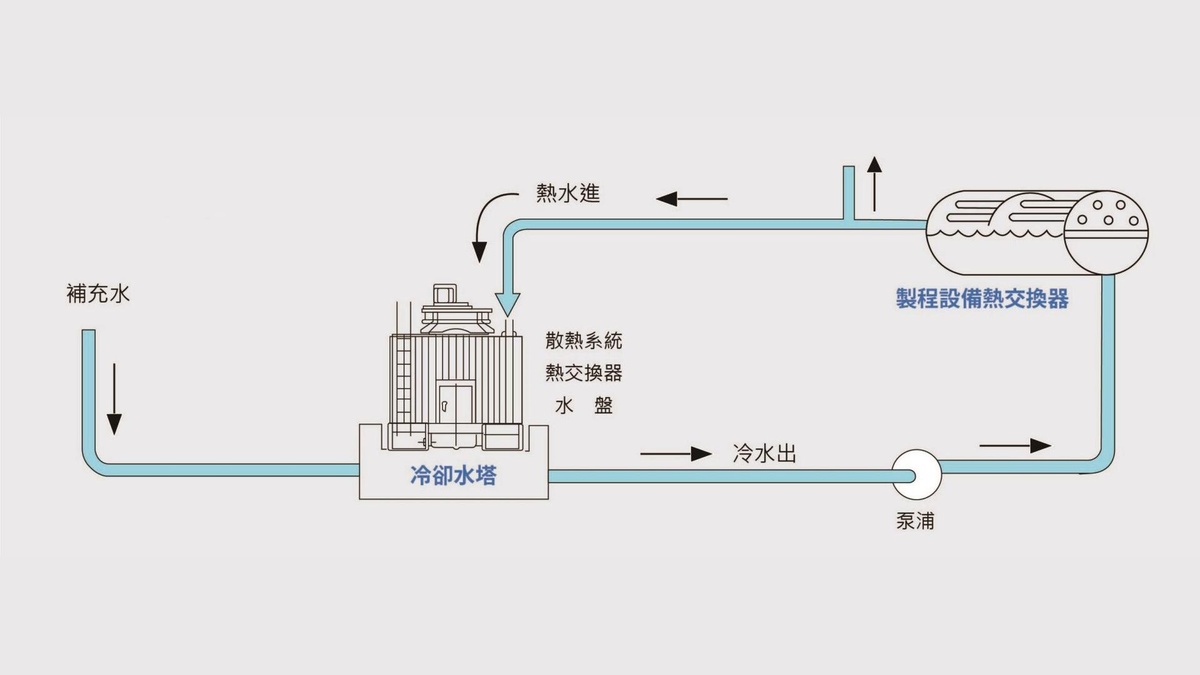
The Production Process of Sintering Technology:
- Sintering: Heat treatment of powder or compact at a temperature lower than the melting point of the components, to increase its strength through metallurgical bonding between particles.
- Packing material: A material in which the compact is embedded to separate and protect it during the pre-sintering or sintering process.
- Presintering: The heat treatment of the compact at a temperature lower than the final sintering temperature.
- Pressure: A sintering process in which uniaxial pressure is applied at the same time as sintering.
- Loose-powder sintering, gravity sintering: The powder is sintered directly without pressing.
- Liquid-phase sintering: Powders or compacts with at least two components are sintered in a state of forming a liquid phase.
- Oversintering: Sintering in which the sintering temperature is too high and the sintering time is too long, resulting in the deterioration of the final performance of the product.
- Undersintering: The sintering temperature is too low and the sintering time is too short so that the product does not reach the required performance.
- Infiltration: A processing method of filling pores in an unsintered or sintered product with a metal or alloy whose melting point is lower than that of the product in a molten state.
- Dewaxing, burn-off: Use heating to discharge organic additives (binders or lubricants) in the compact.
- Mesh belt furnace: A sintering furnace in which parts are continuously conveyed in the furnace by mesh belts generally protected by muffles.
- Walking-beam furnace: A sintering furnace in which the parts placed in the sintering tray are transported in the furnace through a walking beam system.
- Pusher furnace: A sintering furnace in which parts are loaded into a burning boat, and parts are transported in the furnace through a propulsion system.
- Neck formation: Neck-like connections are formed between particles during sintering.
- Blistering: Blistering is formed on the surface of sintered parts due to the violent discharge of gas.
- Sweating: The phenomenon that the liquid phase seeps out during the heat treatment of the compact.
- Sinter skin: During sintering, a surface layer is formed on the sintered part whose properties are different from those inside the product.
- Relative density: the ratio of the density of the porous body to the density of the same component material in the non-porous state, expressed as a percentage.
- Radial crushing strength: The breaking strength of the sintered cylindrical sample is measured by applying radial pressure.
- Porosity: The ratio of the volume of all pores in a porous body to the total volume.
- Diffusion porosity: The pores formed by the diffusion of one component substance into another component due to the Kirkendall effect.
- Pore size distribution: The percentage of pore sizes at all levels in the material is calculated by number or volume.
- Apparent hardness: The hardness of the sintered material measured under specified conditions, which includes the effect of pores.
- Solid hardness: The hardness of a phase or particle or a region of a sintered material measured under specified conditions, which excludes the influence of pores.
- Bubble-point pressure: The minimum pressure required to force gas through a liquid-impregnated article to create the first bubble.
- Fluid permeability: The amount of liquid or gas passing through a porous body in unit time measured under specified conditions.
General Sintering Process:
Sintering can effectively reduce porosity and enhance properties such as strength, electrical conductivity, transparency, and thermal conductivity. In other cases, however, it may be useful to increase its strength but maintain its gas absorbencies, such as in filters or catalysts. During the firing process, the diffusion of atoms will drive the interface between powder particles to disappear. This process will be divided into different stages. The initial stage will form from the neck connection between the powders to the final elimination of small pores.
The driving force for densification is the change in total free energy due to the decrease in surface area and the decrease in surface free energy after the solid-vapor interface is replaced by solid-solid. It forms a new but lower energy solid-solid interface and the total free energy decreases. At the microscopic scale, the material transfer is affected by pressure changes and free energy differences across the surface. The influence of these effects becomes large if the size of the particles is small. The change in energy is much higher when the radius of curvature is less than a few microns, which is one of the main reasons why many ceramic technologies are based on the use of fine-grained materials. For properties such as strength and conductivity, the binding area associated with particle size is the determining factor. The variables that can be controlled for any given material are temperature and initial grain size since vapor pressure is temperature-dependent.
The energy source for solid-state processes is the change in free energy or chemical potential energy between the neck and the particle surface. This energy creates material transfer in the fastest possible way. If the transfer occurs from the particle volume or the grain boundaries between particles, it results in particle reduction and void loss. Pore elimination occurs faster for samples with many uniform sizes and higher porosity, where boundary diffusion distances are smaller. For later parts of the process, boundary diffusion and lattice diffusion from grain boundaries become important. Temperature control is important to the sintering process because grain boundary diffusion and bulk diffusion are heavily dependent on temperature, size, distribution of material particles, material composition, and sintering environment.
Ceramic Sintering Process:
Sintering is part of the firing process of pottery and other ceramic objects. These objects are made of substances such as glass, aluminum oxide, zirconium oxide, silicon dioxide, magnesium oxide, lime, beryllium oxide, and iron oxide. Some ceramic raw materials have a low affinity for water, have a lower plasticity index than clay, and require organic additives in the pre-sintering stage.
The general procedure for making ceramic objects by sintering powders includes:
- Mix water, binder, deflocculant, and unfired ceramic powder uniformly to form a slurry.
- Spray dry the slurry.
- The spray-dried powder is placed into a mold and pressed to form a green body.
- The green body is heated at a low temperature to burn off the binder.
- Sintered at high temperature to fuse the ceramic particles.
By observing the expansion-temperature curve by optical dilatometer thermal analysis, all characteristic temperatures (including phase transition temperature, glass transition temperature, melting point, etc.) occurring during the sintering cycle of a particular ceramic formulation can be easily obtained. Sintering is associated with significant shrinkage of the material, as the glass phase flows once it reaches its transition temperature and begins to incorporate the powder structure and significantly reduce the porosity of the material. Sintering is usually carried out at high temperatures. Additionally, second and third external forces may be used. The second external force commonly used is pressure. Therefore, sintering using only temperature is often referred to as pressureless sintering. Using graded metal-ceramic composites, nanoparticle sintering aids, and monolithic molding techniques, pressureless sintering can be achieved. A variant for 3D shapes is called hot isostatic pressing.
To effectively stack products in the furnace during sintering and prevent parts from sticking together, many manufacturers use ceramic powder separator layers to separate products. Various materials such as alumina, zirconia, and magnesia can be used for these separators. They are divided into different categories by granularity such as fine, medium, coarse, etc. By matching the material and particle size of the separator layer to the product being sintered, surface damage and contamination can be reduced while maximizing the furnace load.
Metal Powder Sintering Process:
Most metals can be sintered. This applies in particular to pure metals produced in a vacuum, which are not subject to surface contamination. Sintering at atmospheric pressure requires the use of shielding gas, usually an endothermic gas. Sintering, followed by reprocessing, can yield a wide range of material properties. Variations in density, alloying, and heat treatment can alter the physical properties of various products. For example, for lower sintering temperatures, Young's modulus En of sintered iron powder remains somewhat insensitive to sintering time, alloying, or particle size in the original powder, but depends on the density of the final product. Where D is the density of the material, E is the theoretical Young's modulus of iron, and d is the theoretical density of iron. Sintering is static while the metal powder may exhibit coalescence under certain external conditions, and still returns to its normal behavior when these conditions are removed. In most cases, as the material flows into the void, the density of the particle assembly increases, resulting in a decrease in the overall volume. The mass movement that occurs during sintering includes the reduction of total porosity through repackaging, followed by material transport due to evaporation and diffusive condensation. In the final stage, the metal atoms move along the crystal boundaries to the walls of the inner pores, redistributing mass from the inner body of the object and smoothing the pore walls. Surface tension is the driving force for this movement.
Spark Plasma Sintering Process:
The SPS process uses pulsed DC to repeatedly energize the material with instantaneous high current, so discharge occurs between particles. Due to the small contact surface between particles, local high temperature can be generated, and plasma, the reduced particle gap is conducive to surface diffusion and boundary defect diffusion, so the powder particle interface is melted and bonded to each other to complete the powder metallurgy sintering process. Due to the high sintering energy, the holding time can be greatly shortened, and the high temperature only occurs in the surface area of the particles, so the grains inside the particles will not grow, which can effectively control the grain size of the sintered body, which is conducive to the microstructure of the sintered body. High-density material sintering. In addition, this technology combines powder forming and sintering in a single process, which does not require pre-forming and does not require any additives and binders.
The parameters affecting the material properties after sintering are temperature gradient, current density, material conductivity, mold wall thickness, current pattern, heating rate, holding time, and sintering pressure. Therefore, the low temperature and short time of spark plasma sintering can greatly reduce the process energy consumption and improve the material process speed. Generally, the bismuth telluride material produced by the smelting process has a smelting temperature of about 550°C or more. The processing time is more than 1 hour, and the nanostructure cannot be formed in the material. Due to the spark plasma sintering technology, the mixed material is nanosized by high-energy ball milling, so the uniformity of the sintered material is better than that of the smelting process material, which can greatly reduces the material loss and thus reduce the development cost.